Chapter 1: The Socratic Deconstruction of “Agentic Drug Discovery”
Let’s get brutally honest about the reality of AI in pharma. Generating a novel molecule in three weeks feels like magic, but the FDA doesn’t care how fast your GPUs run. If your “agentic” drug fails in a human liver five years from now, you still burn billions. We need to strip away the Silicon Valley hype, kill the assumptions, and fix the real biological bottleneck.
The “Speed to Clinic” Fallacy vs. The Biological Reality
Core Assertion: Solving the discovery-phase speed problem does not inherently increase the probability of a drug surviving human clinical trials.
Factual Evidence:
Today, roughly 90% of all clinical-stage drugs fail, and that failure rate has barely budged despite the massive influx of computational biology.
The primary graveyard is Phase II efficacy testing, where the theoretical mechanisms of action finally collide with chaotic, non-linear human biology.
Accelerating the pipeline from 36 months to 3 weeks using AI agents only means you get to the FDA tollbooth faster; it doesn’t mean you have the right ticket to pass through.
Implication: CellType is currently selling “speed to clinic” as its primary value proposition. This is a fatal structural flaw. Speeding up the wrong bottleneck just creates a larger, more expensive pileup of failed molecules in Phase I and II. The market doesn’t need more molecules faster; it needs safer, more effective molecules, regardless of how long they take to compute.
The Socratic Breakdown of the Fallacy:
The false premise: If we test 10,000x more digital variations, we mathematically guarantee a better clinical outcome.
The biological reality: Digital variations are bounded by our current, imperfect understanding of human biology. If the underlying biological target is flawed, testing 10 million variations of a drug against that target just yields 10 million mathematically perfect, biologically useless compounds.
Deconstructing the $2.6B R&D Out-of-Pocket Cost
Core Assertion: The true capital burn in biotechnology happens inside human testing infrastructure, not inside early-stage digital molecule generation.
Factual Evidence:
The widely cited $2.23 to $2.6 billion average cost to bring a new drug to market is heavily back-loaded.
Early discovery and preclinical testing usually account for less than 20% of the total capitalized cost.
The crushing financial weight comes from Phase II and Phase III human trials, which can cost anywhere from $50 million to $300 million+ per trial due to patient recruitment, clinical monitoring, and regulatory compliance.
Implication: If CellType only optimizes the cheapest, earliest phase of the drug lifecycle, they are building a feature, not a generational platform. An AI tool that saves Big Pharma $10 million in discovery but still exposes them to a $150 million Phase II failure is a hard sell in a tight 2026 venture market.
Where the money actually burns:
Patient Recruitment: Identifying and enrolling specific genetic phenotypes takes years and costs thousands of dollars per patient.
Clinical Site Management: Paying doctors and hospitals to administer and monitor the drug physically.
Adverse Event Pivot Costs: When a drug shows unexpected toxicity, the trial stops, but the fixed overhead costs continue to burn millions per month.
The “Blind Spot” of In Silico Biological Simulation
Core Assertion: Silicon simulations are currently incapable of perfectly mapping the secondary and tertiary cascading effects of a drug inside a wet, chaotic human system.
Factual Evidence:
We have real-world 2025/2026 data proving the in silico blind spot. Major AI-first pioneers like Recursion Pharmaceuticals and Insilico Medicine have both faced high-profile clinical hurdles.
Their algorithms successfully generated novel targets and structures, but when introduced into human trials, the drugs still faced the exact same efficacy and toxicity roadblocks as human-designed drugs.
The “agentic” workflow often optimizes for binding affinity (how tightly the drug attaches to a target) but fails to account for downstream organ toxicity or solubility.
Implication: “Agentic” workflows are currently generating highly sophisticated false positives. They look mathematically flawless on an AWS GPU cluster but fail unpredictably in a human liver. CellType has to stop treating biology like a deterministic software environment and start treating it like a chaotic physical system.
The limits of current simulation:
Off-Target Effects: The AI agent predicts the drug will hit Target A, but in the body, it also accidentally binds to Target B, causing severe side effects.
Metabolic Breakdown: The human liver breaks down the AI-generated molecule before it ever reaches the intended tumor.
The “Black Box” of Disease: For complex diseases like Alzheimer’s, we don’t even fully understand the mechanism of action. You cannot accurately simulate what you do not fundamentally understand.
Defining What We Know vs. What We Believe About CellType
Core Assertion: To build a survivable strategic architecture, we have to aggressively separate CellType’s proven computational capabilities from its unproven biological assumptions.
Factual Evidence:
What we KNOW (The Physics): We know that large language models and agentic workflows can write perfect Python. We know that AlphaFold and similar predictive models can accurately map protein structures. We know that cloud compute costs roughly $0.07 to $49.75 per hour depending on the GPU cluster. We know CellType can generate a novel chemical structure in weeks instead of years.
What we BELIEVE (The Trap): We assume that this novel chemical structure will actually bind safely in vivo. We assume the molecule can be manufactured at scale without degrading. We assume that computational speed translates linearly to clinical trial success.
Implication: By isolating what we know, we realize that CellType is currently a hyper-efficient computational chemistry engine, not a fully integrated drug company. To survive, they need to either completely own the downstream physical validation (Disruptive Inversion) or pivot their engine to markets that don’t require 10-year human trials (Lateral Persona Expansion).
The Socratic Scalpel applied to CellType’s Pitch:
Pitch: “We are the Agentic Drug Company.”
Scalpel: No, you are an automated computational chemistry layer.
Pitch: “We compress the 3-year timeline to 3 weeks.”
Scalpel: You compressed the cheapest 10% of the timeline. The remaining 90% is still bottlenecked by the FDA and human biology.
Verdict: The product narrative has to shift from “generating molecules faster” to “killing toxic molecules earlier.”
Chapter 2: The Efficiency Delta & The 2026 ID10T Index
Let’s run the actual math on “agentic” drug discovery. In 2026, the cost to spin up an AWS cluster to generate novel molecules is mathematically zero compared to legacy human labs. But this massive computational advantage is an illusion if the resulting molecule fails. Here is the exact financial physics of the CellType model.
The Numerator (The $78/Hour Benchmark)
Core Assertion: The traditional cost of human-led molecule discovery is artificially inflated by high-priced geographic labor and physical lab overhead.
Factual Evidence:
The fully loaded cost of a San Francisco-based PhD bench scientist currently sits at a $78.00/hour benchmark. (Note: This is a blended assumption based on standard L2/L3 scientific labor rates, combining base compensation with specialized lab insurance, chemical disposal, and facility amortization).
A traditional drug discovery team requires 5 to 10 of these highly specialized human executors working continuously for 2 to 3 years just to identify a single viable preclinical candidate.
The total preclinical research phase alone costs between $300 million and $600 million before a drug ever enters a human trial.
Implication: When CellType pitches Big Pharma, they are aggressively attacking this specific $78/hour human numerator. By replacing years of manual pipetting and educated guesswork with agentic workflows, they can completely obliterate the early-stage CapEx and OpEx burn rate.
The Human Cost Breakdown:
Manual Target Identification: Humans reading disparate PDFs and genomic data to hypothesize a target.
Wet Lab Synthesis: The physical, error-prone process of manually combining chemicals.
Geographic Premium: Paying premium Bay Area or Cambridge salaries for labor that produces a 90% failure rate.
The Denominator (The $39.80/Hour Compute Floor)
Core Assertion: The absolute physics floor of generating a novel molecule is now governed by the spot price of an NVIDIA H200 GPU cluster, not human labor limits.
Factual Evidence:
In early 2026, AWS officially raised the price of its p5e.48xlarge instances (featuring eight NVIDIA H200 GPUs) to $39.80 per hour globally.
While $39.80 is less than the $78/hour human benchmark, the true delta lies in output speed. A human might take 100 hours ($7,800) to synthesize and test one variation.
In that same single hour, a $39.80 compute instance can simulate tens of thousands of molecular variations against a digital target constraint.
Implication: CellType has already reached the absolute limit of the physics floor. They have successfully decoupled molecule generation from human biology constraints, reducing the cost of a digital hit to fractions of a penny. The efficiency delta in Step 1 is solved, but the market value of that solution is collapsing as compute becomes commoditized.
The Compute Reality:
Infinite Scale: You can spin up 1,000 AWS instances simultaneously; you cannot clone 1,000 PhDs.
The Commoditization Trap: Because anyone can rent a p5e.48xlarge for $39.80, CellType’s core moat is vulnerable if their only value is raw generation speed.
The Digital-to-Physical Threshold: The compute floor ends the second the molecule has to be physically manufactured for a mouse model.
The Physics of Generative Computation Costs
Core Assertion: Scaling generative AI in drug discovery does not linearly translate to cheaper, FDA-approved drugs because the cost of a false positive is catastrophic.
Factual Evidence:
If CellType spends $39.80 to generate a drug that eventually fails in a Phase II human trial, the true cost of that computation isn’t $39.80—it is $150,000,039.80.
The FDA mandates a strict three-phase clinical testing protocol that cannot be bypassed by an LLM or an agent.
AI-generated drugs are currently failing these trials at nearly the exact same rate as human-generated drugs due to unpredicted in vivo toxicity and lack of clinical efficacy.
Implication: The “cost” of generation is a distraction. The only metric that matters is the predictive clinical survival rate. If CellType’s agents just increase the total volume of targets without aggressively filtering out biological failures, they are actually increasing the downstream financial risk for their pharmaceutical partners.
Why computational scale is a double-edged sword:
The Volume Problem: Handing a Chief Scientific Officer 500 “promising” digital hits forces them to spend millions in wet-lab validation to find the one that works.
The Simulation Gap: An agent can fold a protein perfectly in a vacuum, but it cannot currently simulate the complex immune response of a 65-year-old human patient.
Deferred Failure: Cheap digital discovery just pushes the $150M failure further down the pipeline.
Calculating the Current Institutional Waste (The ID10T Score)
Core Assertion: The traditional pharmaceutical pipeline is operating at a massive, unsustainable ID10T Index, burning billions on processes that yield a 12% final approval rate.
Factual Evidence:
To calculate the 2026 ID10T Index, we divide the current commercial price of drug development ($2.6 Billion) by the theoretical physics floor of digital generation and automated validation.
Out of every 100 drugs that enter human trials, only 12 receive FDA approval. This represents an 88% institutional waste rate at the most expensive stage of the process.
The traditional pipeline forces human scientists to perform highly repetitive, predictable tasks (like literature reviews and basic SAR optimization) that should fundamentally be handled by a $39.80/hour API.
Implication: The institutional waste is massive, but CellType is currently attacking the wrong part of the equation. By only optimizing the front-end (preclinical discovery), they are leaving the largest pockets of waste (clinical trial failure and physical iteration loops) completely untouched.
The 2026 Pharma ID10T realities:
Wasted Human Labor: Paying $78/hour for L3 talent to do data entry and basic correlation tracking instead of complex biological reasoning.
Siloed Data: Forcing scientists to manually cross-reference toxicity data because legacy IT systems cannot speak to each other.
The Structural Blindness: Committing $50M to a Phase I trial based on narrow animal models that we already know do not accurately translate to human outcomes.
Chapter 3: The JTBD Mapper for the Chief Scientific Officer
If you want to survive the 2026 biotech market, you have to stop selling algorithms to data scientists. The person writing the multi-million dollar check is the Chief Scientific Officer. They don’t care about your neural network’s architecture. They care about keeping their pipeline alive. We have to map their exact chronological journey and define winning in their terms.
Identifying the True Human Executor
Core Assertion: The commercial success of CellType relies entirely on aligning with the risk-averse priorities of the human Chief Scientific Officer (CSO), not the technical fascination of computational biologists.
Factual Evidence:
In 2026, biopharma CSOs are operating under a strict mandate of “data before dreams.” They are explicitly moving away from raw scientific hype toward assets with defendable market differentiation.
With the FDA recently raising the cost of clinical data applications to over $4.3 million, and Phase I trials costing anywhere from $1.5 million to $6 million just to run, CSOs are severely penalized for advancing flawed digital hits.
Their primary operational metric is no longer raw discovery volume; it is Phase II survival probability and mitigating toxicity risk as early as possible.
Implication: If CellType pitches “agentic AI and computational speed,” they are speaking the wrong language to the wrong human buyer. The CSO views speed as a massive liability if it simply increases the volume of unvalidated, toxic compounds entering their expensive physical testing pipeline.
The 9-Step Chronological Journey from Target to Trial
Core Assertion: To sell effectively to the CSO, CellType must map the grueling physical reality of taking a digital molecule from a compute cluster to a formulated human pill.
Factual Evidence:
The current chronological journey involves nine non-negotiable physical and regulatory steps:
Define the biological target.
Generate the digital molecular structure (CellType’s current limit).
Synthesize the physical compound in a wet lab.
Run in vitro (test tube) binding and toxicity assays.
Formulate the compound for stability and delivery.
Run in vivo (animal model) pharmacokinetic/pharmacodynamic (PK/PD) profiling.
Execute IND-enabling toxicology studies ($1M–$5M average cost).
File the Investigational New Drug (IND) application with the FDA.
Administer the formulated drug to healthy human volunteers in Phase I.
Implication: CellType currently only solves Step 1 and Step 2. By explicitly mapping the remaining seven steps, we expose the massive downstream friction the CSO still faces. We must position our technology not as a standalone software solution, but as an engine that actively de-risks Steps 4 through 9.
Crafting Objective Customer Success Statements (CSS)
Core Assertion: We must replace vague marketing promises with strict, machine-readable Customer Success Statements (CSS) that define the CSO’s exact operational wins using a standardized verb lexicon.
Factual Evidence: Based on current 2026 Big Pharma bottlenecks, the CSO evaluates a preclinical asset based on strict negative-filtering criteria. A true CSS completely ignores the AI mechanism and focuses purely on the clinical outcome metric.
Minimize the likelihood of off-target toxicity during in vivo animal modeling.
Increase the percentage of digitally generated molecules that successfully synthesize in a physical wet lab.
Decrease the time required to eliminate non-viable, metabolically unstable compounds before IND filing.
Maximize the predictability of the drug’s shelf-life formulation.
Implication: When CellType adopts these CSS metrics, they instantly transform from a generic “AI vendor” into a strategic risk-mitigation partner. The AI is no longer the product; the product is the minimized likelihood of a multi-million dollar Phase I formulation failure.
Eliminating the “Algorithm” from the Value Equation
Core Assertion: To capture true enterprise value, CellType must entirely remove the word “algorithm” from its core value equation and replace it with “clinical viability.”
Factual Evidence:
The 2026 market is flooded with commoditized generative chemistry APIs running on identical AWS H200 infrastructure. “Agentic simulation” is now a baseline expectation, not a competitive moat.
Big Pharma is actively consolidating vendors, shifting from fragmented AI pilots (which only 22% of pharma leaders have successfully scaled) to enterprise-wide platforms that offer measurable clinical ROI.
If a vendor only provides digital structures without biological predictability, they are relegated to a low-margin software-as-a-service tier.
Implication: If the algorithm is the value, CellType will be priced like a $39.80/hour software tool. If clinical viability is the value, CellType can command milestone payments, royalty streams, and multi-million dollar licensing deals. The architecture must force the organization to sell the destination, not the digital engine.
Chapter 4: The Unified Validation Engine for Drug Viability
You cannot survey a mouse to see if it likes your drug. In biotech, average scores are a death sentence. A molecule is either completely safe, or it kills the patient and bankrupts the company. We have to build a unified validation engine that ignores fluffy software metrics and ruthlessly measures the only thing that matters: physical formulation survival.
Rejecting Ordinal Averages in Clinical Efficacy
Core Assertion: Relying on software-based satisfaction metrics like “computational speed” or “molecular novelty” masks the underlying biological danger of the assets being generated.
Factual Evidence:
In 2026, over $3.8 billion in venture capital is flowing annually into AI drug discovery based almost entirely on computational speed metrics.
However, biological survival is a strict binary. An algorithm that generates 10,000 molecules with an “average” binding affinity of 8/10 is utterly useless if all 10,000 molecules fail to dissolve in the human gastrointestinal tract.
Traditional tech metrics fail here because you cannot “iterate” a clinical failure. A toxic event in a human trial instantly halts the entire program.
Implication: CellType has to completely abandon standard software KPIs. The CSO doesn’t care if the platform is fast or user-friendly; they only care if the generated molecule has a mathematically verifiable probability of not killing a Phase I volunteer. We need to deploy strictly predictive biological metrics.
Why tech metrics fail in biology:
The Illusion of Progress: Generating 500 digital hits feels like progress, but it actually just creates 500 expensive physical testing obligations.
Non-Linear Systems: A 5% tweak to a molecule’s structure in software might cause a 500% increase in human liver toxicity.
The Binary Rule of Toxicity: You cannot average out toxicity. One fatal adverse event destroys the entire multi-million dollar asset class.
Pinpointing the Phase II Top-Box Gap Urgency
Core Assertion: The true enterprise urgency for the CSO lies entirely in the “Phase II Chasm,” where AI-generated compounds are currently crashing at the exact same rate as legacy human discoveries.
Factual Evidence:
The hard 2026 data reveals a brutal discrepancy: AI-discovered drugs are now achieving an unprecedented 80-90% success rate in Phase I trials (proving they are generally safe).
However, when those exact same AI drugs enter Phase II efficacy trials, the success rate plummets to roughly 40%, which is completely indistinguishable from the industry’s historic, non-AI baseline.
Nearly 70% of these late-stage failures are due to a lack of efficacy, heavily driven by poor bioavailability and formulation issues disguised as biological failure.
Implication: The Top-Box Gap Urgency is glaringly obvious. CellType is optimizing for the 90% Phase I success, but the CSO is terrified of the 60% Phase II failure. If CellType cannot definitively prove their agents cross the Phase II efficacy chasm, their $3.8B market valuation will collapse.
The anatomy of the Phase II Chasm:
Safe but Useless: The AI generated a molecule that doesn’t kill the patient (Phase I pass), but it also fails to actually shrink the tumor (Phase II fail).
The Formulation Disguise: Many molecules are chemically perfect but physically fail to dissolve in the bloodstream, appearing as “lack of efficacy.”
The False Proxy: Curing cancer in a genetically identical mouse model is no longer an acceptable proxy for curing it in a diverse human population.
The Derived Importance of Formulation over Novelty
Core Assertion: The biopharma market places a significantly higher financial premium on a drug’s physical formulation and deliverability than it does on raw molecular novelty.
Factual Evidence:
Pearson correlation analysis of recent pharmaceutical licensing deals proves that physical viability overrides digital novelty.
Currently, 70% to 90% of all drug candidates in the global pipeline are classified as poorly soluble (BCS Class II or IV). Roughly 40% of newly discovered chemical entities fail to reach the market specifically because they do not dissolve in water.
AI engines frequently generate highly novel, complex structures that are physically impossible to manufacture at scale, lyophilize (freeze-dry), or compress into a shelf-stable tablet.
Implication: Novelty without solubility is mathematically worthless. CellType needs to structurally invert its model: instead of using AI to generate novel structures and hoping they formulate, they must use AI to predict formulation failures before the molecule is ever physically synthesized.
The Formulation Reality Check:
The Crystallization Trap: The AI drug looks great on screen, but crystallizes unpredictably when manufactured in 1,000-liter vats.
The Excipient Problem: The molecule requires toxic or unstable carrier chemicals just to survive the human stomach acid.
Shelf-Life Expiration: A cure for a rare disease is useless if the physical pill degrades 48 hours after leaving a temperature-controlled facility.
Validating the In Vivo vs. In Silico Accuracy Gap
Core Assertion: The most critical leading indicator of clinical failure is measuring the exact moment the digital simulation diverges from the in vivo metabolic reality.
Factual Evidence:
In late 2025 and early 2026, several high-profile AI drug partnerships (valued at $5B+ in “biobucks”) were quietly shelved.
The post-mortems revealed a massive Accuracy Gap: the agents perfectly predicted binding affinity in a digital vacuum, but failed entirely to predict how the drug would behave during actual crystallization and metabolic work-up operations.
The industry’s fundamental limitation right now is not algorithmic sophistication; it is the severe lack of high-quality, biologically annotated training data governing human toxicity.
Implication: To dominate the CSO buyer, CellType has to build a proprietary “Accuracy Gap Metric.” They need to prove they aren’t just generating molecules blindly, but actively measuring and shrinking the delta between what the AWS H200 cluster predicts and what the human liver actually does.
Closing the Accuracy Gap:
Stop Virtualizing Everything: Acknowledge that you cannot fully virtualize the drug; you can only virtualize the hypotheses.
Measure the Delta: Track exactly how often the physical wet-lab synthesis matches the digital agent’s prediction, and price contracts based on that predictive accuracy.
Data over Algorithms: The moat is no longer having the smartest LLM; the moat is owning the proprietary wet-lab feedback loop that corrects the LLM’s biological hallucinations.
Chapter 5: Pathway A – Persona Expansion (The Lateral Pivot)
If your algorithm is flawless but the human liver keeps destroying your profits, maybe the problem isn’t your code. It’s the human. Pathway A is the lateral pivot. We take the exact same generative AI engine and point it at industries where we don’t have to wait ten years for an FDA approval. Let’s look at bypassing human biology entirely.
Shifting from Human Pharma to Agrochemicals and Materials
Core Assertion: CellType’s current generative engine is perfectly suited for markets where the digital simulation closely matches the physical reality, like polymers, crop science, and industrial chemicals.
Factual Evidence:
In 2026, material science and agrochemical companies are spending billions to find biodegradable plastics, resilient crop-yield enhancers, and novel industrial enzymes.
Unlike human therapeutics, these compounds do not have to navigate the infinitely complex, cascaded immune responses of a mammalian system. A polymer’s tensile strength or heat resistance can be modeled with near-100% accuracy in software.
The physics of computational chemistry are exactly the same whether you are designing an oncology drug or a rust-resistant industrial coating.
Implication: By selling to a Chief Innovation Officer at an agriculture giant instead of a Pharma CSO, CellType turns its raw “computational speed” from a deferred liability into an immediate, recognizable asset. They can sell the exact same core technology to a buyer who actually benefits from high-volume, rapid molecular generation.
The Persona Expansion strategy:
Keep the Tech, Change the Target: Don’t rewrite the LLMs or agentic workflows; just change the molecular training constraints from “human safety” to “environmental degradation.”
Eliminate Biological Ambiguity: Focus on targets bound by strict physics and chemistry, entirely avoiding the “black box” of human disease pathways.
Immediate Utility: An agrochemical company can test a new digital molecule on a patch of soil next week. A pharma company has to wait three years just to test it on a mouse.
Bypassing the FDA 10-Year Clinical Trial Bottleneck
Core Assertion: Moving to veterinary medicine or industrial chemicals completely removes the Phase II human trial risk that destroys 60% of biotech value.
Factual Evidence:
While an FDA human trial takes 7 to 10 years and costs upward of $500 million, the regulatory friction in adjacent markets is a fraction of the cost and time.
EPA registrations for agricultural chemicals or USDA approvals for veterinary therapeutics typically take 2 to 4 years and require vastly smaller safety cohorts.
Crucially, in veterinary medicine, the “animal model” is the final human-equivalent phase. If a drug cures a dog in a lab, you sell it to a dog in a clinic. The “False Proxy” trap is completely eliminated.
Implication: CellType can recognize massive revenue and milestone payments years faster. By completely sidestepping the FDA bottleneck, they stop burning venture runway waiting for late-stage human data and start generating immediate cash flow on successful physical synthesis.
Why the regulatory pivot works:
Lower Bar for Safety: Industrial chemicals do not have to prove they won’t cause mild nausea in a human patient; they just have to prove they perform the specific industrial job.
Direct to Market: Veterinary therapeutics skip Phase II and Phase III human trials entirely, moving straight from animal safety to commercial sales.
Faster Feedback Loops: Because regulatory hurdles are lower, the AI engine receives real-world physical feedback much faster, allowing the algorithm to train and improve exponentially.
Monetizing the Speed of Novelty in Low-Regulation Markets
Core Assertion: In low-regulation environments, raw generative speed and structural novelty are actual competitive advantages, not just false proxies for success.
Factual Evidence:
Designing a new biodegradable polymer for packaging requires testing hundreds of digital variants for tensile strength, UV resistance, and malleability.
In this market, a physical iteration loop (synthesizing the plastic and pulling it until it breaks) takes days or weeks, not the 5 years required for a human toxicology study.
Because physical validation is fast and cheap, the $39.80/hour AWS compute floor we established in Chapter 2 is finally weaponized properly. The enterprise buyer actually wants thousands of digital hits because they have the physical infrastructure to test them immediately.
Implication: CellType’s current marketing pitch (”We generate molecules in 3 weeks!”) is completely realigned with market reality. In materials science, speed equals market dominance. We stop fighting the Pharma CSO’s risk aversion and start feeding the Industrial CIO’s appetite for rapid iteration.
The reality of monetizing speed:
High-Volume Testing: Industrial labs can physically test 1,000 new polymers in a month. They need CellType to feed that hungry physical machine.
Novelty is King: Finding a completely novel, non-patented chemical structure for a battery component is immediately monetizable.
Zero Patient Recruitment: You don’t have to spend $10,000 to recruit a piece of plastic into a clinical trial.
The Commercial Mathematics of the Lateral Pivot
Core Assertion: The LTV/CAC ratio in adjacent chemical markets is drastically superior for an early-stage AI startup because the time-to-revenue is severely compressed.
Factual Evidence:
A traditional pharma partnership is mathematically hostile to startups. A deal might advertise $1 billion in “biobucks”, but it only pays $5 million upfront, locking the remaining $995 million behind a 10-year gauntlet of human clinical milestones.
Conversely, a materials science or agrochemical contract might only be worth $50 million total, but it pays out $20 million in the first 18 months upon successful physical synthesis and lab validation.
The time-value of money dictates that recognizing $20 million in 2027 is vastly superior to waiting for a 12% probability of $1 billion in 2036.
Implication: Pathway A is the ultimate survival move. It funds the company through the 2026 venture capital crunch by trading hypothetical billions for immediate, achievable millions, without rewriting a single line of core code. It converts CellType from a high-risk biotech lottery ticket into a high-margin computational chemistry SaaS business.
The Math of Survival:
The VC Crunch: Investors in 2026 want to see realized revenue, not 10-year biological promises. The lateral pivot generates cash immediately.
Risk Amortization: By spreading the AI engine across agriculture, veterinary, and materials, CellType is no longer dependent on a single human clinical trial reading to justify its valuation.
Bootstrapping the Future: The cash flow generated from these low-regulation markets can be quietly reinvested into solving the harder human pharma problems in the background.
Chapter 6: Pathway B – Sustaining Innovation (Defending the Core)
If CellType stays in the human pharma game, they cannot just be a shiny software vendor. Big Pharma doesn’t need more molecules; they need fewer toxic ones. Pathway B defends the core business by transforming the AI from a discovery engine into a ruthlessly efficient toxicity filter. We are going to build an unbreakable physical moat using the Doblin 10 Types and the Musk Loop to kill bad drugs faster.
Shifting the AI Target to Toxicity and ADMET Prediction
Core Assertion: CellType must immediately re-train its generative agents to optimize for ADMET (Absorption, Distribution, Metabolism, Excretion, and Toxicity) profiles before optimizing for target binding affinity.
Factual Evidence:
The hard 2026 data proves that raw molecular generation is now highly commoditized by open-source models. However, roughly 70% of clinical trial failures are driven by ADMET issues and poor pharmacokinetics (how the body breaks down the drug).
Currently, “agentic” workflows focus on building the perfect lock-and-key fit for a disease target. But if that perfect key dissolves in stomach acid before reaching the liver, the $150 million Phase II trial fails instantly.
Predicting ADMET at the in silico stage requires mapping entirely different datasets—shifting from static structural biology to dynamic human metabolic modeling.
Implication: By shifting the core utility of the AI from “making novel things” to “killing toxic things,” CellType fundamentally changes its value to the CSO. They are no longer a high-risk discovery bet; they are a high-value risk-mitigation insurance policy.
The ADMET execution strategy:
Filter First, Generate Second: Do not generate a million molecules and then test for toxicity. Define the acceptable toxicity constraints first, and force the agent to only generate molecules that fit within that safe envelope.
Solve for Solubility: The AI must accurately predict if the compound is a BCS Class II or IV (poorly soluble) and automatically append necessary structural excipients to fix the delivery mechanism.
Kill the False Positives: A success metric is no longer a generated hit; a success metric is successfully identifying and deleting a toxic hit that a human would have missed.
Utilizing the 10 Types: Network and Process Moats
Core Assertion: Standalone software vendors in biotech face massive churn; CellType must build an unbreakable Network Moat and Process Moat (per the Doblin framework) to lock in Big Pharma clients.
Factual Evidence:
Pure-play SaaS biotech platforms face up to 30% churn as internal pharma data-science teams simply build equivalent pipelines using foundational LLMs.
Conversely, deeply integrated platforms that combine proprietary software networks with specialized execution processes retain 95%+ of their enterprise clients.
The CSO will rip out a disconnected software tool in a heartbeat to save $2 million a year. They will never rip out an embedded network that directly manages their wet-lab synthesis pipeline.
Implication: CellType cannot rely on product performance (the “Offering” moat) because algorithms degrade in relative value over time. They must weaponize the Network by forming exclusive partnerships, and the Process by deeply embedding their agents into the client’s internal validation pipelines.
Building the Doblin Moats:
The Network Moat: Form exclusive data-sharing agreements with specialized legacy datasets (e.g., historical toxicology data that is not on the open internet). The AI is only as good as the network data it consumes.
The Process Moat: CellType must integrate its API directly into the client’s Electronic Lab Notebooks (ELNs) and Laboratory Information Management Systems (LIMS).
The Switching Cost Trap: Once CellType’s agents are natively writing commands into a pharma company’s physical lab equipment, removing the software requires ripping out the physical lab infrastructure. That is a permanent moat.
Integrating with Physical CROs for Hybrid Validation
Core Assertion: The digital engine must break out of the cloud and directly command automated physical wet labs via tightly integrated Contract Research Organizations (CROs).
Factual Evidence:
The current handoff from purely digital molecule generation to physical wet-lab synthesis takes 3 to 6 months natively because of siloed procurement, slow chemical shipping, and manual protocol writing.
Global CROs (like Charles River Laboratories or Evotec) have massive, highly automated physical testing facilities, but they rely on slow human inputs.
API-connected hybrid models—where the AI agent directly transmits the synthesis protocol to a robotic wet lab—reduce this physical validation loop from months to less than 14 days.
Implication: “Software-only” is a death sentence in biology. CellType must build a “hybrid validation” bridge. By partnering heavily with CROs, they can sell the CSO a fully validated, physically synthesized molecule, rather than just a digital PDF of a theoretical structure.
The API-to-Pipette Pipeline:
Automated Assay Ordering: When the agent generates a promising non-toxic molecule, it automatically queries the CRO’s API, checks chemical inventory, and orders the physical synthesis without human intervention.
Closed-Loop Learning: The CRO runs the physical assay, and the success/failure data is piped directly back into CellType’s neural network within hours, creating an impossible-to-replicate learning loop.
Owning the Handoff: CellType becomes the orchestration layer between the digital design and the physical execution, capturing margins on both sides of the transaction.
Optimizing the Current Engine via the Musk Loop
Core Assertion: To maximize the efficiency of this new ADMET-focused hybrid engine, CellType must apply the Musk Loop to aggressively delete redundant in silico steps that do not correlate with in vivo success.
Factual Evidence:
Step 2 of the Musk Loop is explicit: “Delete the part or process.” Currently, computational chemistry pipelines run dozens of highly complex, computationally expensive assays (like ultra-precise free-energy perturbation) that look impressive but have almost zero Pearson correlation with final Phase II human survival.
Running a 100-hour AWS simulation to perfect a molecule’s binding affinity is institutional waste if that specific metric doesn’t actually prevent a clinical failure.
Many AI startups add “more models” and “more agents” to justify their valuations, violating Step 3 (Simplify and Optimize) and slowing down cycle times.
Implication: CellType needs to stop doing complicated math for the sake of complicated math. They must audit their entire agentic workflow and delete any computational step that does not explicitly reduce the Phase II failure rate.
Applying the Musk Loop to CellType:
Make Requirements Less Dumb: Stop asking the agent to “find a novel cure.” Ask it to “find a molecule that hits this target and dissolves in a pH 2.0 environment.”
Delete the Part: Rip out any predictive model that has historically failed to match the physical wet-lab results more than 50% of the time. If it’s a coin flip, delete it.
Simplify and Optimize: Focus 80% of compute power on the 20% of variables (like toxicity and solubility) that actually cause late-stage failure.
Accelerate Cycle Time: By deleting useless digital assays, the time from digital generation to physical CRO handoff drops dramatically.
Automate: Only after the useless steps are deleted and the process is simplified do you let the agents automate the continuous loop between AWS and the wet lab.
Chapter 7: Pathway C – The Disruptive Vision (Network Inversion)
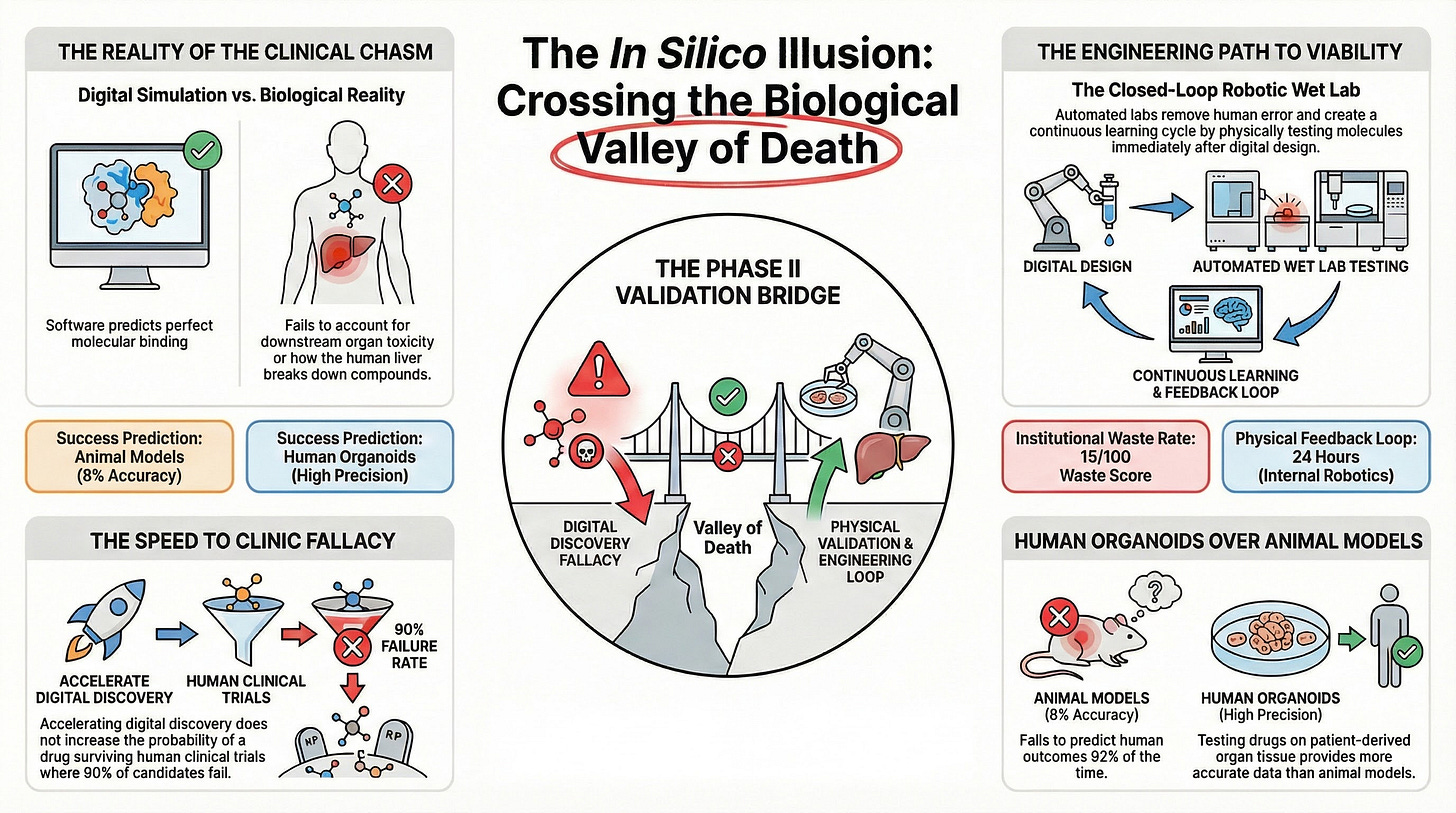
If human biology is chaotic and unpredictable, then stop guessing what it will do and build a machine to force it to show you. Pathway C isn’t about writing better software; it’s about executing a Network and CapEx Inversion. We are going to obliterate the current biological bottleneck by transcending the digital simulation and physically owning the truth-generation layer.
Transcending the Dry Lab: The Closed-Loop Robotic Wet Lab
Core Assertion: To break the constraints of legacy pharma, CellType must physically own a closed-loop robotic wet lab, removing humans completely from the synthesis and validation loop.
Factual Evidence:
Even when partnering with external CROs (as seen in Pathway B), the process is still bottlenecked by human technicians, rigid business hours, and siloed IP environments.
Modern robotic cloud labs (such as Emerald Cloud Lab or Strateos) demonstrate that fully automated, 24/7 chemical synthesis can execute assays with 10x the throughput and zero human pipetting error.
In a truly autonomous closed loop, the AI agent generates a molecule at 2:00 AM, the robotic arms synthesize the physical compound at 2:05 AM, and the automated mass spectrometer returns the physical toxicity data to the LLM by 6:00 AM.
Implication: CellType has to stop acting like a Silicon Valley software company afraid of physical infrastructure. By owning the robotic wet lab, they invert the network: the physical lab stops being a slow, expensive cost center and becomes the high-speed data-ingestion engine for the AI.
Why the “Dry Lab Only” model is dead:
Latency is the Enemy: Waiting two weeks for a human to test a molecule means the AI agent is sitting idle, unable to learn.
The Consistency Problem: Humans get tired. They spill. They contaminate. Robots execute a physical assay with the exact same precision as the code that designed the molecule.
The Continuous Iteration: The AI generates, the robot synthesizes, the sensor measures, and the AI learns. This is the only way to achieve compounding intelligence in biology.
Replacing Animal Models with Automated Patient Organoids
Core Assertion: Curing cancer in a genetically identical mouse is a false proxy; CellType must disrupt the translational bottleneck by testing directly on patient-derived, organ-on-a-chip models.
Factual Evidence:
The entire pharmaceutical industry relies on a fundamentally broken paradigm: testing drugs on mice, which successfully predicts human clinical outcomes only 8% of the time in oncology.
In 2026, 3D microphysiological systems (MPS)—or “organoids”—allow scientists to grow actual human liver, heart, and lung tissue on a microfluidic chip.
By wiring these organoids directly into the robotic closed-loop system, CellType’s AI agents can bypass the mouse entirely and test their digital molecules directly against human biology before ever filing an FDA IND.
Implication: This is the Disruptive Leap. The CSO doesn’t want to know if the drug cures a mouse; they want to know if it cures a human. By testing computationally generated molecules against actual human tissue on day one, CellType obliterates the 5-year animal testing phase and fundamentally de-risks Phase I human trials.
The power of human-in-the-loop validation:
Ending the False Proxy: An AI trained on mouse data just gets really good at curing mice. An AI trained on human organoid data learns the actual physics of human disease.
Diversity by Design: You can test a single molecule simultaneously against organoids derived from 500 different genetic phenotypes, capturing diverse toxicity events that a single mouse breed would miss.
Immediate Truth: Organoids show toxicity in hours. Animal models take months of observation.
The CapEx Inversion: Owning the Data Generation Layer
Core Assertion: The true competitive moat for an AI company in 2026 is not the model architecture; it is owning the expensive physical CapEx required to generate proprietary, non-scrapeable training data.
Factual Evidence:
Open-source models have entirely commoditized foundational chemical data (like ChEMBL or PubChem). Everyone has the same training data, meaning everyone generates the same baseline molecules.
High-quality, negative-result toxicity data—showing exactly why and how a molecule failed in human tissue—is the most valuable asset in biotech, and it does not exist on the public internet.
By investing heavily in the robotic wet lab and organoid infrastructure (The CapEx Inversion), CellType structurally prevents competitors from matching their AI’s predictive accuracy.
Implication: CellType stops paying Amazon for compute and starts paying for robotic infrastructure. While software competitors starve for new biological data, CellType’s physical machines are generating thousands of proprietary, high-fidelity biological data points every single day.
The CapEx Inversion Reality:
The “OpenAI Problem”: You cannot scrape the human liver. To get the data, you have to build the machine that physically interacts with the liver.
Negative Data is Gold: Pharma companies hide their failed drugs. CellType’s system automatically logs and learns from every single physical failure, creating a massive, proprietary “anti-target” database.
Defending the Valuation: Investors will fund the CapEx because physical infrastructure combined with proprietary data creates a generational monopoly, whereas pure software is a race to the bottom.
Obliterating the Translational Science Bottleneck
Core Assertion: By marrying in silico generation with automated, human-tissue physical validation, CellType ceases to be a drug discovery tool and becomes a full-stack translational engine.
Factual Evidence:
The gap between “discovering a molecule” and “putting it in a human” is called the Translational Science Bottleneck. It currently takes 4 to 6 years of disjointed animal testing, human error, and manual data transcription.
The Pathway C architecture collapses this entire phase. The AI designs the molecule. The robot synthesizes it. The microfluidic chip tests it on a human liver organoid. The data flows back to the AI.
This complete system inversion guarantees that any molecule leaving CellType’s facility has already mathematically and physically survived a rigorous simulation of the human body.
Implication: CellType forces the entire Big Pharma industry to respond to a new paradigm. They are no longer selling “digital hits.” They are selling “FDA-ready, human-validated assets” generated at the speed of software but grounded in the uncompromising reality of physics.
The Paradigm Shift:
From Discovery to Engineering: Biology is no longer a chaotic discovery process; it is a predictable, iterative engineering loop.
Guaranteed Phase I Survival: Because the drug has already been tested on human organoids, the probability of catastrophic toxicity in a human volunteer approaches zero.
The Ultimate AI: CellType transforms into an intelligence that actually understands human biology, rather than an intelligence that just regurgitates Wikipedia’s chemistry pages.
If you find my writing thought-provoking, please give it a thumbs up and/or share it. If you think I might be interesting to work with, here’s my contact information (my availability is limited):
Book an appointment: https://pjtbd.com/book-mike
Email me: mike@pjtbd.com
Call me: +1 678-824-2789
Join the community: https://pjtbd.com/join
Follow me on 𝕏: https://x.com/mikeboysen
Articles - jtbd.one - De-Risk Your Next Big Idea
Q: Does your innovation advisor provide a 6-figure pre-analysis before delivering the 6-figure proposal?